A New Preputial Structure
Senior Lecturer in Pathology, Faculty of Health Studies, Auckland University of Technology
Textbooks and papers referring to penile function state that the source of penile sensation is solely the glans and often justify the existence of the prepuce by stating it protects the sensitive
glans. These statements are contrary to the neuro-anatomical and physiological facts accumulated over more than a century. This study examined the findings of Taylor, et al., that the prepuce is the primary sensory platform of the penis, and describes a new preputial structure.
Looking back, it is astounding that the complex nature of the prepuce (or the fact that it is not a simple fold of skin) was not described until 19911 and that this did not appear in the mainstream medical media until 1996.2 When Dr. John Taylor faced family enquiries, he asked himself: What exactly is removed by circumcision?
A total absence of an answer to that question from the medical literature was the motivation for his investigation. The reason why this had never been asked, during a century of extensive study of the male genitalia and increasing application of circumcision, remains a mystery. No special techniques, or dissections, or equipment were initially required — just an observant eye and scientific curiosity.
When Taylor and his colleagues examined 22 prepuces in males aged 22 to 58 years,2 they discovered that the classical description, which simply divided the prepuce into outer and inner (or mucosal) areas,3 was incomplete. The outer area is a continuation of the shaft skin, up to the rim of junction with the mucosa at the opening of the prepuce at rest, with all its characteristics of form and mild sensitivity. On the other hand, the mucosa, that inner preputial skin from the junctional or transitional zone to the sulcus where it continues as the mucosal covering of the glans, he found to be more sophisticated than had been previously described. Just inside the junctional rim of the prepuce at the dorsal aspect is a transverse band of ridges 10-15 mm wide. Beyond this ridged band, the remainder of the mucosa is smooth (15-30 mm wide dorsally) — the smooth zone. The ridged band is continuous with the frenulum, being a radial band over about 60% of the penile diameter — the dorsal and lateral aspects — then turning distally to form a V
shape in the ventral aspect whilst smoothly reducing its width to merge with the frenulum. This continuity is the reason why the ridged band is also known as the frenular band. Under microscopic examination, the entire mucosa is invested with Meissners corpuscles (the nerve endings which mediate low-threshold and fine-touch sensation) which are heavily concentrated in the projections of the dermis that rise into the ridged band and in the frenulum. These corpuscles are innervated by thick myelinated axons of the epicritical sensory system. A recent quantitative study has shown mean axon bundle counts in the prepuce of 17.9 bundles/mm ventrally, 8.6 bundles/mm laterally and 6.2 bundles/mm dorsally.4 As each bundle contains tens of axons, the nerve supply to the prepuce is obviously very substantial.
Another feature of the inner prepuce not previously described prior to Taylors reports is the extensive plexus of venules and capillaries that are collected into the large superficial veins of the outer prepuce and penile shaft skin. These superficial veins begin to divide as they approach the prepuce and have frequent anastomoses especially in the outer prepuce and distal shaft skin. The anastomoses in the prepuce have sometimes been termed radial veins
because, in some individuals, they are circumferential. From the junctional rim, the many smaller veins divide frequently as they run towards the sulcus to form a dense plexus of venules and capillaries. This vascularity was noted by Taylor as being pronounced in the ridged band and often seen as a bright flush.2
In most genitally intact males, some core branches of the superficial veins do not divide further on crossing the inner prepuce. These medium-sized veins continue unchanged to run under the corona to form at least part of the drainage of the glans surface. This is contrary to a report by Breza, et al., that describes the superficial veins as draining the shaft skin and prepuce only.5 This otherwise excellent and extensive paper is flawed by not reporting the state (intact or circumcised) of the cadaver penises examined. It is reasonable to suppose that, in California, all, or the majority, were circumcised and thus could not present the normal anatomy of the superficial veins. Clearly, the volume of blood gathered by these veins, and their consequent prominence, is greater than could be involved solely in drainage of this skin. Furthermore, the superficial veins become more prominent with erection, suggesting that they may be carrying extra blood from the glans portion of the corpus spongiosum. It may be postulated that: (1) as circumcision interrupts this venous circuit and (2) as superficial veins are not restored in the great majority of preputial remnants, the change in the color of the glans from a venous purple in genitally intact males to the pink tint of the penile shaft in circumcised males is due to a dramatic change in skin circulation in the circumcised penis.
The other major feature of the prepuce noted by Taylor, which it shares with the shaft skin, is a layer of smooth muscle bundles.2 This peripenic muscle has been known since at least 1907 and is the continuation of the dartos muscle of the scrotal skin.6 In the shaft skin, these muscle bundles are oriented in mixed directions,7 but they become specifically oriented in the prepuce to add to its complexity. In the outer prepuce, the bundles are aligned longitudinally to mediate the length of that portion of skin. At the tip of the prepuce (i.e., in the junctional zone), the muscle bundles are radially oriented to form a kind of sphincter which tends to close the orifice when the penis is flaccid.6 Together with abundant elastic fibers, this muscle layer gives the prepuce a very snug fit over the glans. In the absence of constriction of the preputial orifice (a preputial stenosis or true pathological — as opposed to physiological — phimosis,
which can cause back-flow of the urinary stream), all extraneous matter (including urine, sand, etc.) is prevented from gaining access to the preputial space.
Finally, a comment on what is probably the longest enduring myth in anatomy. To the present day, textbooks still assert the presence of glands in the mucosa of the sulcus, especially on both sides of the frenulum, despite the existence of these so-called Tysons glands being rejected by many European investigators as far back as 1884.8 The problem has arisen because one of the findings of Tysons paper of 1699 on the orangutan was interpreted as applying to humans, which it does not.9 No actual description of them in humans exists and a number of modern investigations have failed to find any glands in the inner prepuce or sulcus.10-13 My colleague, Christopher Cold, and I have also failed to find any glands in this region. The 1904 comment of Keith and Shillitoe best sums up this myth: It shows the strength of the faith of anatomists that they have for nearly two centuries described glands which have not, and never had, an existence.
8 It remains to observe that smegma is epithelial debris shed from the mucosa suspended in a proteinaceous fluid;12,14 Taylor and I believe this fluid to be a transudate — extracellular fluid that leaks out from between cells that have no tight junctions.15
Some months ago, whilst rereading the report of Taylor, et al., I realized that their description and illustrations of the inner prepuce and ridged band were confined to the dorsal aspect.2 The frenular attachment of the ridged band was described, but there was no other mention or an illustration of the ventralaspect.
The ridged band is not circumferential over the ventral aspect, since it curves distally to merge with the frenulum, as noted above. This means that the smooth zone is progressively reduced as the median raphe is approached until it disappears at the mid-line. As the smooth zone is reduced, a corresponding zone of the inner prepuce appears proximally between the ridged band and the circumferential junctional rim. Because the length of the inner prepuce actually increases as the median raphe is approached due to the curving of the corona towards the meatus to form the ventral cleft in the glans, the mucosal area of the prepuce is also greater in the ventral aspect. Thus, in the ventral prepuce, there is an undescribed zone that occupies about 20-40% of the mucosa.
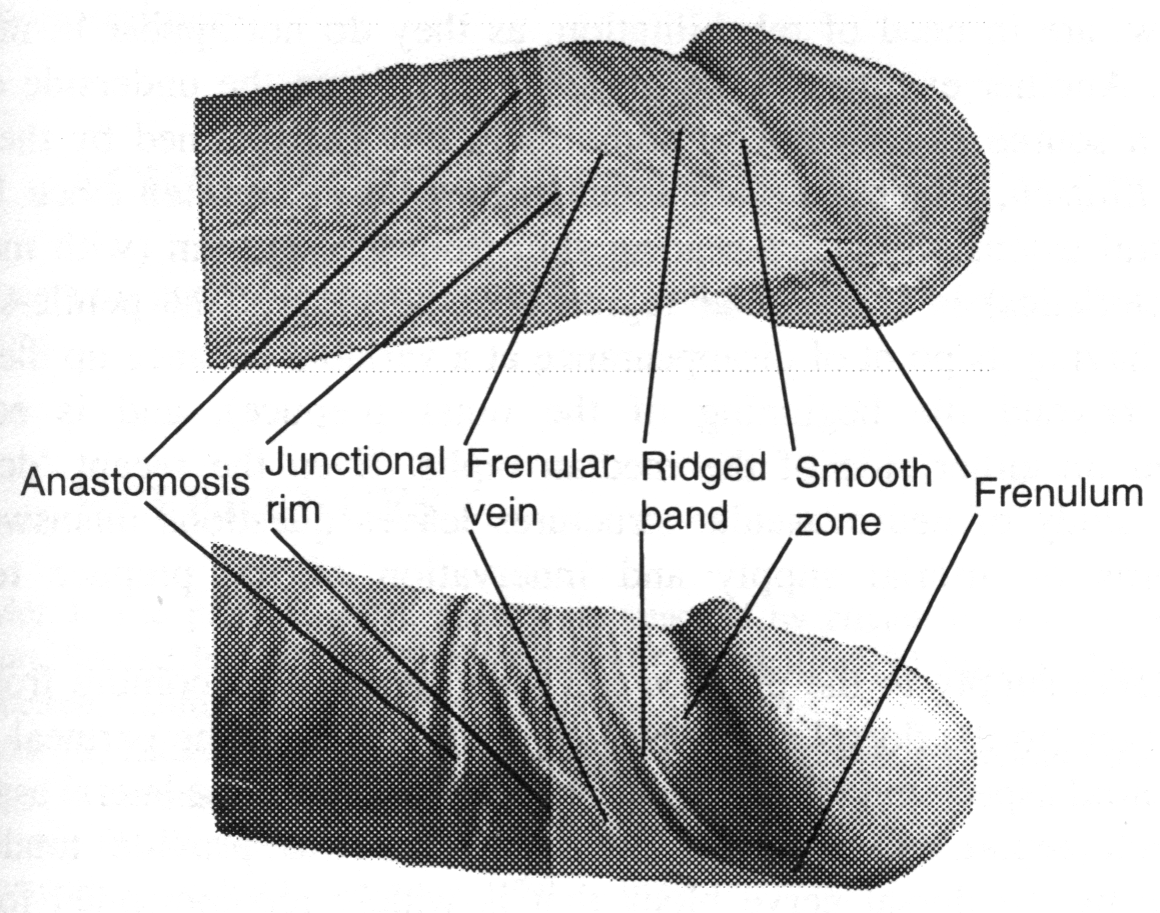
I propose the name frenular delta for this zone, because of its shape and its association with the frenulum. It may be described as that preputial mucosa of triangular shape proximal to the ridged band in the ventral aspect, having the frenulum at its point, its sides defined by the ridged band and as its base the junctional rim. It presents as the greater area of the mucosa ventrally and does not include the ridged band which is a zone in its own right. The median raphe runs longitudinally through its mid-line to become the frenulum and bisects the delta into two equal halves. In males with a short prepuce, the ridged band lies immediately inside the junctional rim at rest and against the glans in a variable position according to the preputial length. The frenular delta in these individuals does not extend beyond the lateral aspects. On the other hand, males with a long prepuce forming an akroposthion (the tubular and tapered part of the prepuce that extends beyond the glans penis) have their ridged band lying over the glans tip at rest. In this situation, the frenular delta base extends beyond the lateral aspects in a ribbon-like band as the extra dorsal strip of mucosa between the junctional rim and the ridged band. The frenular delta is noted by men as the most sensitive area of their penis, especially in the mid-line nearest the frenulum, and the frenulum itself.
Two of the medium veins of passage
described above are of relevance here: regardless of the pattern and numbers of superficial veins, two medium-sized branches curve from each side of the outer prepuce to course through the frenular delta ever closer to the median raphe, eventually to lie on either side of the frenulum before disappearing into its depths. In this way, they appear eventually to flank the frenular artery to form a triplet analogous to the two dorsal arteries and deep dorsal vein of the penile shaft. For these two veins, I propose the name frenular veins. After careful observation of a large number of intact penises, I have not found any that lack these veins and would also observe that they are almost never seen in circumcised penises. There is no observable division of these veins into smaller venules before they disappear into the sides of the frenulum, so we may assume that they drain the area of the glans supplied by the frenular artery (i.e., the immediate surroundings of the cleft and meatus).
Our knowledge of penile anatomy is far from complete. Some of the early findings on gross anatomy, such as the peripenic muscle mentioned in the review, are in need of rehabilitation, as they do not appear in modern literature. Another example is the presence of hair on the underside of the penis — a source of concern for some men; this is explained by the area scroti of Klatsch, which does not seem to have been reported since 1914.6 This ventral extension of hair-bearing skin from the scrotum (with most of its characteristics) is a triangular region with its base at the penile-scrotal junction, having its point of disappearance at a variable distance up the shaft (but not beyond the beginning of the outer prepuce), and is equally distributed on either side of the median raphe. Even the recent detailed anatomy
study of neurovascular structures leaves questions unanswered:5 the patterns of arterial supply and innervation of the prepuce remain unknown.
The nerve supply to the prepuce has been reported as coming from the dorsal nerves (to the dorsal and lateral aspects) and from the perineal nerve (to the ventral aspect and frenulum) with some overlap in the lateral aspect.16 This recent report explains the frequent failure to provide total skin anesthesia by the dorsal nerve block that is widely recommended for and used in circumcision.17 How these nerves reach the prepuce from their deeper sites remains unanswered and leaves us with a physiological conundrum: because the whole skin of the penis is uniquely mobile, any direct branches of nerves to the surface would be broken by this extensive longitudinal and radial movement. Nerve bundles must, therefore, enter from either end of the tubular skin, as do the superficial blood vessels. Ironically, circumcision provides an important clue, since nerves running down the skin to the prepuce from the proximal end at the penile base would be severed by that excision of a large circumferential segment of skin, leaving the preputial stump and frenulum denervated. As circumcised men often describe the preputial and frenular remnants as highly sensitive, it seems apparent that innervation of the prepuce must be retrograde, i.e., entering the skin from the distal end and progressing back towards the base. Moreover, nerves regenerating after surgery seldom, if ever, cross the scar, as it is an impenetrable barrier to the delicate growth cones sprouting from the proximal nerve stump which often form neuromata at the scar boundary. Neuromata have been seen recently by Cold in microscope sections of circumcision scars.18
Given the ventral nerve bundle counts and the universal accounts of its sensitivity, which it shares with the frenulum, the frenular delta must have impressive sensory resources. Tracing of preputial nerve pathways and quantitative studies of nerve endings should provide evidence to confirm these observations. The neurological differences from the female equivalents also need to be more thoroughly investigated.19 That the so-called G-spot
of males could have been ignored by neuro-anatomical studies to the present day is extraordinary.
The author is grateful to Martin Novoa and Christopher Price for helpful discussions and suggestions.
Glands.Ectopic sebaceous glands and papillomatosis penis. Arch Dermatol 1969;99:31-6.
The Circumcision Information and Resource Pages are a not-for-profit educational resource and library. IntactiWiki hosts this website but is not responsible for the content of this site. CIRP makes documents available without charge, for informational purposes only. The contents of this site are not intended to replace the professional medical or legal advice of a licensed practitioner.
© CIRP.org 1996-2026 | Filetree | Please visit our sponsor and host:
IntactiWiki.