MRC News (London), Pages 20-23. Summer 1998.
MRC News (London), Pages 20-23. Summer 1998.

We all experience pain at some time. Fortunately the sensation is usually brief but in some cases it persists. Increasing understanding in recent years of how pain is processed in the brain has brought exciting breakthoughs in pain-killing treatments. But this research has focused on adults. When it comes to babies, things are quite different.
Adults can describe and quantify their pain, report of the effectiveness of a pain-killer and develop strategies for coping with persistent problems. But this is not true of infants and children. Babies cannot tell you about pain and even school children find it hard to describe.
Until about ten years ago, it was assumed infants simply did not experience pain. Babies born prematurely, would undergo essential but traumatic medical or surgical procedures without painkilling drugs. Even young children's analgesic requirements were considered to be much lower than adults.
The problem was that little was known about the development of somatosensation - how the brain acquires its ability to detect body stimulation - and their was no information at all on the state of the nerve pathways that signal pain in newborn babies.
In recent years, however, new biological knowledge has started to shed light on how our nerve pathways develop and reveal a very different picture.
Our laboratory was the first to investigate how pain signaling, or nociception, develops in the newborn, and our first step was to establish whether the infant nervous system is capable of reacting to harmful stimulation in an organised predictable and measurable way.
When adults and older children are hurt, specialised sensory nerves known as nociceptors send signals to the spinal cord and brain which prompt the body into automatic awarewess of pain. Since they cannot talk about it, it is very difficult to asses newborn infants' perception of pain, but it is possible to measure the protective reactions which, we know from adults, exactly parallel the sensation of pain.


To test the level of pain processing in the human infant spinal cord, we focused on the reflex which causes the body to automatically draw back from a painful stimulus (Figure 1). We proposed that infants exposed to the kind of stimuli they might experience in intensive care or surgery would either respond weakly because their pain system is too immature to function properly, or they would react in a similar way to adults but perhaps less efficiently or completely.
What we did not predict was that they would respond quite differently to adults, and that their somatosensory system would involve unique, short-lived signalling pathways not found in the mature nervous system.
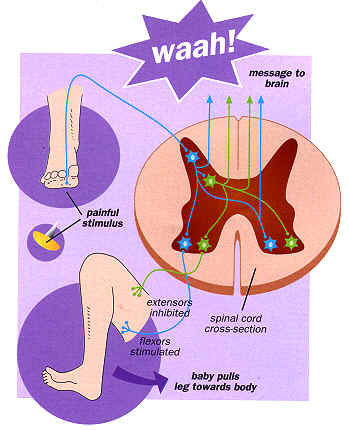
Our work showed that three key aspects of sensory processing in the spinal cord underlie responses to pain in the newborn.
First, an infant's spinal sensory nerve cells are more excitable than an adult's. This makes their spinal reflex response to a harmful stimulus much greater and more prolonged.
Second, individual sensory nerve cells in newborns are linked to much larger areas of skin - or receptive fields than in adults. This means reflex responses can be triggered from a larger body area. Receptive fields combine to form a 'map' or image of our body surface in our brain and enable us to locate a stimulus in a particular part of our body. In newborns, it appears, the larger receptive fields result in a less precise or 'smeared' body map compared to adults.
Lastly, where adults only produce pain reflexes when they encounter harmful stimulation, newborns respond less selectively and produce the same reflex even to a light touch.
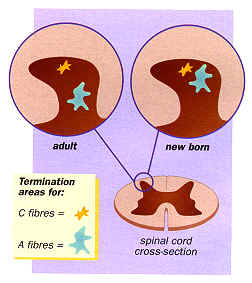
We have revealed some of the mechanisms which underlie these sensory features. For example, we found that in newborns the sensory nerve fibres that communicate non-harmful touch - known as A
fibres - end is a different part of the spinal cord from adults.
The spinal cord cells in this area control several pain mechanisms such as those which cause the pain response to increase when exposed to a repeated stimulus, and those which trigger genes to produce chemicals in response to pain. In adults, the cells are only connected to pain transmitting C fibres
(see Figure 2), but in newborns, because of the different wiring, they can be triggered by non-painful, A
fibre stimulation.
These nerve fibres from the brainstem start to grow down the spinal cord early in fetal life, but they do not extend branches into the spinal cord for some time, and do not function fully until soon after birth. This means the premature baby can not benefit from the natural pain-killing system which in adults dampens down pain messages as they enter the central nervous system.
Do these discoveries mean that the newborn infant's spinal cord transmittts a different pain signal to the cortex to that of the adult? We think so.
Much of the immaturity that we have found suggests newborns have a spinal sensory system that is more sensitive than the adult system. This may have a developmental advantage, encouraging weak nerves cells junctions to strengthen through plenty of activity. But it may also mean newborns have increased false alarms.
They are also more likely to be more unpredicatable in their sensory responses, since their spread-out central terminals and large spinal cord cell receptive fields are likely to lead to poorer placing of stimuli and less selective responses.
How this affects the brain and an infant's awareness of pain is unclear, since very little is known about the processing of pain in the brain. It is important to remember that these findings relate only to spinal cord pain mechanisms which are one step in the pain pathway, and that the actual experience or awareness of pain requires brain activity which we believe develops some time after birth along with memory, anxiety and other cognitive brain functions.
However, the evidence is building up that the importance of adequate analgesia for infants and children goes beyond their immediate comfort and wellbeing. Reports in clinical and psychological literatue indicate early injury or trauma can have long-term consequences on sensory or pain behavior that extend into childhood and beyond.
In adults, for example, MRC researcher Clifford Woolf has shown that when a sensory nerve is damaged or chronically inflamed, the nociceptive mechanisms change and the nervous system no longer relies on signals from the site of the injury to trigger pain. Instead, pain is generated by altered activity within the spinal cord and the brain itself is much harder to tolerate or to control. (see MRC News Issue 67).
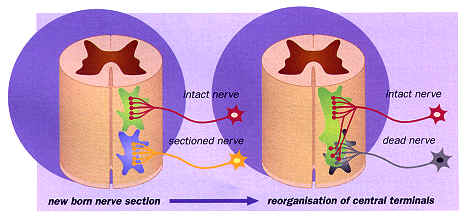
We and others have established that the developing nervous system is even more vulnerable to injury than in adults and that changes to the pathways induced shortly after birth can become permanent. This is because newborn nerve damage not only results in the death of sensory nerve cells, but causes other sensory nerve terminals to sprout extensively and occupy areas normally exclusively devoted to the damaged nerve.
These new sprouts form inappropriate connections with spinal nerve cord cells in areas far outside their normal termination region, causing the nervous system to become permanently distorted (See Figure 3). Even a simple skin wound at birth can trigger changes which result in the area becoming over-supplied with sensory nerve terminals. This leaves the area hypersensitive to touch long after the wound has healed (see Figure 4).
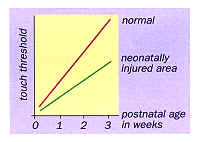
Although in the short term these changes could be called a useful compensatory device to make up for the loss of sensory input from an area, the long-term effects of a permanent alteration in the body's sensory mapping may be detrimental.
Our aim now is to investigate the biological basis for this adaptability of the nervous system and the long-term consequences of early injuries. What we find will be of major importance for clinicians and babies in their care.
Maria Fitzgerald is Professor of Developmental Neurobiology at the Thomas Lewis Pain Research Centre, Department of Anatomy and Developmental Biology,
University College London and Director of the Children Nationwide Paediatric Pain Group at the
Institute of Child Health. She trained with Professor Patrick Wall at University College London.
MRC News is available free and published quarterly by the Medical Research Council, 20 Park Crescent, London W1N 4AL, Telephone 0171-636 5422
The Circumcision Information and Resource Pages are a not-for-profit educational resource and library. IntactiWiki hosts this website but is not responsible for the content of this site. CIRP makes documents available without charge, for informational purposes only. The contents of this site are not intended to replace the professional medical or legal advice of a licensed practitioner.
© CIRP.org 1996-2026 | Filetree | Please visit our sponsor and host:
IntactiWiki.